2011-2012
Course Information
On this page, you will find notes, PowerPoint presentations, instructions for assignments, homework assignments, and other information pertinent to the curriculum.
_________________________________________________________________
PAST UNITS
__________________________________________________________________
Applications of Aqueous Equilibria
April 10 - April 27th
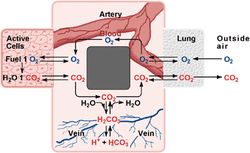
This unit brings acid-base equilibrium into acid/base reactions. We will investigate titration reactions and buffers as well as solubility of various ionic solids.
This unit brings acid-base equilibrium into acid/base reactions. We will investigate titration reactions and buffers as well as solubility of various ionic solids.
Answer to Free Response Problems1. a. Ka = [H3O+][F-]/[HF] b. 1.7 x 10E-2 M c. .0040 mol
d. 0.15 M e. pH=3.32 2. a. 6.7 x 10E-5 M b. OCl- + H2O <--> HOCl + OH- Kb = 3.2x10E-7 c. pH = 10.28 d. 10 millimoles e. pH = 1.18 Lesson Plans
|
Helpful Handouts, Videos, and Websites
|
Electrochemistry and Thermodynamics

April 30th - May 4th
We will quickly visit the electrochemical cells on April 30th and May 1st. Both galvanic (voltaic) and electrolytic cells will be demonstrated. Then we will learn about the interrelationship of enthalpy, entropy, and Gibbs free energy. These thermodynamic concepts help scientists determine whether a reaction will occur. The second law of thermodynamics will be discussed.
We will quickly visit the electrochemical cells on April 30th and May 1st. Both galvanic (voltaic) and electrolytic cells will be demonstrated. Then we will learn about the interrelationship of enthalpy, entropy, and Gibbs free energy. These thermodynamic concepts help scientists determine whether a reaction will occur. The second law of thermodynamics will be discussed.
Lesson Plans
|
Helpful Videos, Handouts, and Websites
|
Acids and Bases

March 29th to April 11th
We will continue with chemical equilibrium and how it applies to acid/base chemistry. Acids and bases are common materials found in nature and in industry. Many reactions would not occur without specific acidic or basic conditions. The reason we have stomach acid is so the enzymes can work to digest our food.
We will continue with chemical equilibrium and how it applies to acid/base chemistry. Acids and bases are common materials found in nature and in industry. Many reactions would not occur without specific acidic or basic conditions. The reason we have stomach acid is so the enzymes can work to digest our food.
Lesson Plans
|
Helpful Handouts, Websites, and Videos
|
Chemical Equilibrium

March 22th - March 29th
We will begin our three chapter long study of chemical equilibria. The concept of chemical equilibria will explain percent yield, will compare and contrast reactions that go to "completion" and those that are easily reversed. Le Chatlier's Principle helps us predict what happens to a chemical system when a stress is applied - especially important in climate studies.
We will begin our three chapter long study of chemical equilibria. The concept of chemical equilibria will explain percent yield, will compare and contrast reactions that go to "completion" and those that are easily reversed. Le Chatlier's Principle helps us predict what happens to a chemical system when a stress is applied - especially important in climate studies.
Helpful Handouts, Videos, and WebsitesChapter 13 notes
Le Chatelier Notes Solving Equilibrium Problems LeChatelier's Simulation - Complex Ion Formation Homework |
Chemical Kinetics
March 8th to March 20th
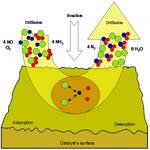
Chemical kinetics involves determining the rates of reactions (with or without catalysts). Reaction kinetics can only be determined experimentally. Our bodies use enzymes (catalysts) to speed up the digestion of our foods. Many industrial chemical production facilities need catalysts to increase rates of reactions and product yield. This will be a "quick" study (pun intended).
Chemical kinetics involves determining the rates of reactions (with or without catalysts). Reaction kinetics can only be determined experimentally. Our bodies use enzymes (catalysts) to speed up the digestion of our foods. Many industrial chemical production facilities need catalysts to increase rates of reactions and product yield. This will be a "quick" study (pun intended).
Lesson Plans
|
Helpful Handouts, Videos, and Websites |
Properties of Solutions

February 27th - March 7th
This unit investigates homogeneous mixtures and their properties. Solubility of solids and gases, miscibility of liquids will be explained and tested. We will learn that adding a salt to water increases the boiling point and decreases the freezing points of the water. Applications are endless in that the world is made of more solutions and mixtures than pure substances. The physical properties of the solutions in always concentration dependent and composition dependent. We will make ICE CREAM and play with Coke and Mentos. How cool is that?
This unit investigates homogeneous mixtures and their properties. Solubility of solids and gases, miscibility of liquids will be explained and tested. We will learn that adding a salt to water increases the boiling point and decreases the freezing points of the water. Applications are endless in that the world is made of more solutions and mixtures than pure substances. The physical properties of the solutions in always concentration dependent and composition dependent. We will make ICE CREAM and play with Coke and Mentos. How cool is that?
Lesson Plans
|
Helpful Handouts, Videos, and Web Sites
|
ATOMIC THEORY DEVELOPMENT AND NUCLEAR CHEMISTRY

Introduction/Review of Atomic Theory Development - Chapter 2
Review of Nuclear Chemistry - Chapter 18 Below are PowerPoints, homework assignments, lesson plans, etc. that relate to this first unit in 2011-2012.
|
QUANTUM MECHANICS

Quantum Mechanics - Unit 2
Chapter 7 PowerPoints, handouts, homework assignments, lesson plans, etc.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Chemical Bonds: General Concepts
Chapter 8
This unit is extremely important in understanding molecules, stereo-chemistry, and reactions. Stereo-chemistry is the study of how the three-dimensional arrangement of atoms in a molecule influences the chemical and physical properties of that molecule. There are important biochemical applications of stereo-chemistry. There may be a more modern word but I like the sound of it. Good websites for visualizing molecular geometry
See them all at once
VIMEO - interesting video This link is similar to the way I teach molecular geometry - MHHE
|
Covalent Bonding: Orbitals
Chapter 9 - October 19th -26th
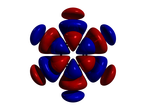
This chapter continues with Unit 3. It goes into greater depth with orbitals, hybridization, and molecular orbitals. Students should begin to see the complexity of sharing electrons within molecules. The content is applicable to the development of medicinal drugs and gene therapy. Hybridization of Atomic Orbitals Model
This a a great simulation of the hybridization of atomic orbitals in the formation of molecules. MHHE
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Stoichiometry
Chapter 3 - October 28th - November 17th
In this chapter, we will review stoichiometry of compounds and reactions. This content applies to all future content areas and it is absolutely necessary to master its content to succeed in subsequent lessons. Topics will include empirical and molecular formulas, mole ratios, formula mass, percent yield, and limiting reactants. Applications are limitless.
We will do our first AP Labs in this unit - various Gravimetric Analysis experiments
In this chapter, we will review stoichiometry of compounds and reactions. This content applies to all future content areas and it is absolutely necessary to master its content to succeed in subsequent lessons. Topics will include empirical and molecular formulas, mole ratios, formula mass, percent yield, and limiting reactants. Applications are limitless.
We will do our first AP Labs in this unit - various Gravimetric Analysis experiments
Report for the Metal Carbonate Lab (click to see the instructions)
The report should include the following sections: title, purpose, data table (measurements only, no calculations), calculations (show work) - include percent error in the calculations, and analysis.
The analysis is the most important part of the lab. Discuss your results, how you determined the answer with the data collected, justify your work using known stoichiometric concepts, and explain your error appropriately. Due two days after the completion of the lab. |
Composition of a Mixture Lab
The report should include all data, calculations, and results as well as an analysis of your results. The analysis should include a justification of your calculations using stoichiometric principles and an appropriate error discussion. Please do not include purpose, procedure, or materials. Due by Monday, November 28th.
|
|
Chapter 3 Homework Assignment
|
Videos, Animations, etc. to help with stoichiometry
Reactions in Solution - Chapter 4
Solutions

Chapter 4 Sections 1 - 3
November 18 - 30 This is an extension of Chapter 3 where we begin to investigate reactions in solutions. In the first part of the chapter, we will discuss various ways to express solution concentration. We work with, drink, or make solutions everyday. Beer's Law Lab Titration Lab - Standardizing a NaOH solution. Solutions PowerPoint Lesson Plans for this unit
Homework
Predicting Products of Reactions
December 14th - 21st
We will use a different text book for this unit. It is more comprehensive than the Zumdahl book we are using. In this section, we will go over reactions and the typical products formed. Th AP Chemistry test always requires that students be able to do this. Students will practice using past test questions. Lesson Plan |
Types of Chemical Reactions

Chapter 4 Sections 4 - 10
December 1 - 13. The rest of Chapter 4 focuses on recognizing what is happening in a reaction, being able to predict the outcome and write a balanced equation for that reaction. Your body is an incredible chemical factory. Redox titration Lab Reactions PowerPoint Study Guide
Helpful Handouts, Web Pages, and Videos - (click to view the documents or video)
|
Gas Laws

January 3rd - January 17th
This unit will include all the gas laws, kinetic molecular theory, stoichiometry, and deviation from ideal behavior. There will be one lab and a few demonstrations. The lab will use the ideal gas laws to predict the molar mass of a gas. Gas laws have widespread applications including atmospheric chemistry, hydrothermal venting (geysers), and volcanic eruptions. This unit will be followed by a midterm exam on stoichiometry. Lesson PlansHelpful Handouts, Videos, and Web PagesLab Report Instructions_A Gaseous Tale Project - on Project page
|
Liquids and Solids

February 7th - February 17th
This unit covers the properties of solids and liquids - the other states of matter. We will answer the question "What makes a solid a solid and what makes a liquid a liquid?" The answer lies in the bonding and intermolecular forces. The physical reality of solids and liquids is EXTREMELY important to our planet and life in general. Lesson PlansHomeworkHelpful Handouts, Videos, and Web SitesSolids PowerPoint
|
Thermochemistry

January 25th - February 6th
In this unit we are introduced to the concept of enthalpy, the measurements of heats of reaction, heat of formation. The evolution and absorption of heat is present in everyday life. Our bodies maintain a temperature well above ambient temperature, we cook our food using heat, climate change is a "hot" topic at the moment. Knowledge of thermochemistry can give anyone a perspective on heat. |
|
