AP CHEMISTRY 2012-2013 UNITS
CURRENT UNIT
~~~~~~~~~~~~~~~~~~~~~~~~~
Thermodynamics and Electrochemistry
April 29-May 2nd
This unit is the culmination of types of reactions, atomic theory, equilibrium and thermodynamics. It is very straightforward but it does require the understanding of many basic concepts we have covered previously.
Lesson Plan
Flashcards for Thermochemistry
Good Overview Website
Good Website
Good Electrochem Websites
This unit is the culmination of types of reactions, atomic theory, equilibrium and thermodynamics. It is very straightforward but it does require the understanding of many basic concepts we have covered previously.
Lesson Plan
Flashcards for Thermochemistry
Good Overview Website
Good Website
Good Electrochem Websites
_______________________________
PAST UNITS
_______________________________
Acid/Base Chemistry

April 1-April 25th
This unit is the culmination of reactions, solutions, stoichiometry, and equilibrium concepts and applications. Students will learn that solutions are not neat and multiple reactions and equilibrium conditions contribute to the true nature of the solution. The problems are multi-stepped and require switching between stoichiometry of reactions and stoichiometry of equilibria. Recognizing the major species in solution is absolutely key to understanding what is happening in solution.
Lesson Plan Chapter 14
Lesson Plan Chapter 15
Websites for Titrations
This unit is the culmination of reactions, solutions, stoichiometry, and equilibrium concepts and applications. Students will learn that solutions are not neat and multiple reactions and equilibrium conditions contribute to the true nature of the solution. The problems are multi-stepped and require switching between stoichiometry of reactions and stoichiometry of equilibria. Recognizing the major species in solution is absolutely key to understanding what is happening in solution.
Lesson Plan Chapter 14
Lesson Plan Chapter 15
Websites for Titrations
Chemical Equlibrium

March 18th - March 27th
Many chemical reactions are reversible. Under the appropriate conditions, the reverse reaction will occur when enough product has been formed. Eventually an equilibrium occurs when the rate of the forward reaction equals the rate of the reverse reaction. Both reactions continue but there is no measureable change in concentrations of the reactants and products - a dynamic equilibrium is established.
Updated Lesson Plans
Basics of Chemical Equilibria
Cobalt Chloride Equilibrium Simulation
Chemical Equilibrium Computer Lab
LeChatelier's Principle
Many chemical reactions are reversible. Under the appropriate conditions, the reverse reaction will occur when enough product has been formed. Eventually an equilibrium occurs when the rate of the forward reaction equals the rate of the reverse reaction. Both reactions continue but there is no measureable change in concentrations of the reactants and products - a dynamic equilibrium is established.
Updated Lesson Plans
Basics of Chemical Equilibria
Cobalt Chloride Equilibrium Simulation
Chemical Equilibrium Computer Lab
LeChatelier's Principle
Chemical Kinetics

March 4th to March 15th
Students will relate rate of reactions to several factors, study rate laws, use experimental data to determine rate laws, determine a rate law experimentally, and witness the effect of a catalyst. Rates of reactions are very important to biological and geological processes.
Lesson Plans
Web Pages
Students will relate rate of reactions to several factors, study rate laws, use experimental data to determine rate laws, determine a rate law experimentally, and witness the effect of a catalyst. Rates of reactions are very important to biological and geological processes.
Lesson Plans
Web Pages
Chapter 10: Solids and Liquids

January 30th - February 11th
The condensed states of matter behave very differently than gases. Gases seem ephemeral but solids and liquids are easily seen, touched, and tasted. We sometimes take them for granite. : ) In this unit, we will investigate the differences among all the physical states of matter and determine the relationship between their physical properties and the forces that hold the solids and liquids together.
Updated Lesson Plans Dipoles-Dipole Interactions
Hydrogen Bonding London Dispersion Forces
Vapor Pressure Website Liquids PowerPoint Solids
Types of Solids Comprehensive Review
The condensed states of matter behave very differently than gases. Gases seem ephemeral but solids and liquids are easily seen, touched, and tasted. We sometimes take them for granite. : ) In this unit, we will investigate the differences among all the physical states of matter and determine the relationship between their physical properties and the forces that hold the solids and liquids together.
Updated Lesson Plans Dipoles-Dipole Interactions
Hydrogen Bonding London Dispersion Forces
Vapor Pressure Website Liquids PowerPoint Solids
Types of Solids Comprehensive Review
Chapter 6: Thermochemistry

January 16th - January 28th
This topic is COOL or hot . . . . Most physical and chemical changes involve significant changes in internal energy of substances. Many processes will gain or lose energy, sometimes as heat. Student's will learn to apply Hess's Law to determine the energy change of a reaction without doing any measurements. This unit along with the Gas Law unit is helpful in understanding weather. Kinetic Molecular Theory will be expanded to explain the transfer of heat from a hot object to a cooler one.
Lesson Plans PowerPoint Slides Hess's Law
Links to Websites Intro to Thermochem William Rankin Story
This topic is COOL or hot . . . . Most physical and chemical changes involve significant changes in internal energy of substances. Many processes will gain or lose energy, sometimes as heat. Student's will learn to apply Hess's Law to determine the energy change of a reaction without doing any measurements. This unit along with the Gas Law unit is helpful in understanding weather. Kinetic Molecular Theory will be expanded to explain the transfer of heat from a hot object to a cooler one.
Lesson Plans PowerPoint Slides Hess's Law
Links to Websites Intro to Thermochem William Rankin Story
Gas laws

January 2nd - January 14th
Gases behave very differently than solids and liquids but very predictably under most conditions. In this unit, we will learn about the physical properties of gases and the mathematical laws that apply to their behavior.
Lesson Plans Effusion and Diffusion Websites
Project Assignment GasLaw review KMT
Gases behave very differently than solids and liquids but very predictably under most conditions. In this unit, we will learn about the physical properties of gases and the mathematical laws that apply to their behavior.
Lesson Plans Effusion and Diffusion Websites
Project Assignment GasLaw review KMT
predicting products of reactions

December 17 - December 21
Students will learn how to determine the products of various types of reactions.
Lesson Plans PowerPoint
Students will learn how to determine the products of various types of reactions.
Lesson Plans PowerPoint
REACTIONS IN SOLUTION

November 30th to December 14th
The chemistry of solutions is very important to life, the Earth, and industry. There are an untold number of different reactions that occur in solutions. It is essential to understand the nature of all reactions but especially those that occur in aqueous solutions. If you can master aqueous reactions, you can master any type of reaction.
Lesson Plans Study Guide to Chapter 4 Links to Balancing Redox Equations
The chemistry of solutions is very important to life, the Earth, and industry. There are an untold number of different reactions that occur in solutions. It is essential to understand the nature of all reactions but especially those that occur in aqueous solutions. If you can master aqueous reactions, you can master any type of reaction.
Lesson Plans Study Guide to Chapter 4 Links to Balancing Redox Equations
Hybrid Orbitals and Molecular Orbital Theory
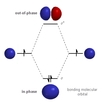
October 17th to October 29th
This unit continues to explain molecular geometry by using new models from atomic orbitals that participate in covalent bonding. The energy of the shared electrons changes and the possible locations also change as electrons are attracted to two nuclei instead of one. Two models are presented in Chapter 9 - Hybridization of atomic orbitals which uses the LE model and a newer model called Molecular Orbital theory which was developed using the same concepts and similar mathematical considerations as Schrodinger's wave equation.
Click on the following to open the documents or websites
Lesson Plans and Homework Assignments Websites
Hybridzation of Atomic Orbitals Sigma and Pi Bonds Animation Molecular Orbital Theory
This unit continues to explain molecular geometry by using new models from atomic orbitals that participate in covalent bonding. The energy of the shared electrons changes and the possible locations also change as electrons are attracted to two nuclei instead of one. Two models are presented in Chapter 9 - Hybridization of atomic orbitals which uses the LE model and a newer model called Molecular Orbital theory which was developed using the same concepts and similar mathematical considerations as Schrodinger's wave equation.
Click on the following to open the documents or websites
Lesson Plans and Homework Assignments Websites
Hybridzation of Atomic Orbitals Sigma and Pi Bonds Animation Molecular Orbital Theory
BONDING
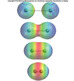
OCTOBER 2 nd - OCTOBER 15 th
This unit which covers Chapter 8 is extremely important to understanding reactions and molecules. It will also reinforce atomic theory and periodicity from Chapter 7. Chemical bonding is directly related to the physical and chemical properties of compounds, metals and multi-atom elements. We will use the information in this chapter in virtually all subsequent chapters.
Click on the following to open the documents
Lesson Plan and Homework Assignments Websites Ionic Bonding PowerPoint
Covalent Bonding PowerPoint Molecular Geometry Perioidc Kingdom Questions Part III
This unit which covers Chapter 8 is extremely important to understanding reactions and molecules. It will also reinforce atomic theory and periodicity from Chapter 7. Chemical bonding is directly related to the physical and chemical properties of compounds, metals and multi-atom elements. We will use the information in this chapter in virtually all subsequent chapters.
Click on the following to open the documents
Lesson Plan and Homework Assignments Websites Ionic Bonding PowerPoint
Covalent Bonding PowerPoint Molecular Geometry Perioidc Kingdom Questions Part III
Modern Atomic Theory
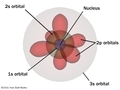
Sept 14th - Sept 21st
In this unit we will review the experiments that lead to the development of atomic theory. Orbital diagrams and electron configuration as well as quantum numbers will be discussed. Periodicitiy will be the next unit.
Lesson Plans Homework PowerPoint #1 PowerPoint #2 PowerPoint #3
In this unit we will review the experiments that lead to the development of atomic theory. Orbital diagrams and electron configuration as well as quantum numbers will be discussed. Periodicitiy will be the next unit.
Lesson Plans Homework PowerPoint #1 PowerPoint #2 PowerPoint #3
Nuclear Chemistry

Sept 10 - 14th
We will start from the inside and work our way outward. This unit will include the importance of the nucleus in the grand scheme of things, why nuclei decay, how they decay, and what happens when they decay. We will discuss the experiments that support the current know model of the nucleus. It will be quick and mostly review.
Nuclear HW Lesson Plans Nuclear PowerPoint
We will start from the inside and work our way outward. This unit will include the importance of the nucleus in the grand scheme of things, why nuclei decay, how they decay, and what happens when they decay. We will discuss the experiments that support the current know model of the nucleus. It will be quick and mostly review.
Nuclear HW Lesson Plans Nuclear PowerPoint

